 GreenPlus Medical
GreenPlus Medical
 GreenPlus Medical
GreenPlus Medical
 Medical Supply Transparent Film IV Cannula Dressing for Wound Care
Medical Supply Transparent Film IV Cannula Dressing for Wound Care
 Medical Grade Wound Transparent Dressings Disposable Waterproof Medical PU IV Cannula Dressing
Medical Grade Wound Transparent Dressings Disposable Waterproof Medical PU IV Cannula Dressing
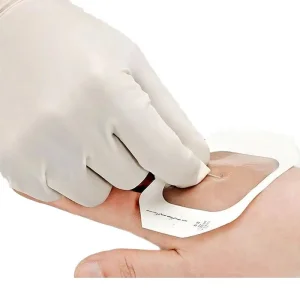 Manufacturer Medical Transparent IV Cannula Film Fixation Wound Dressing Sterile Waterproof PU Self Adhesive Dressings
Manufacturer Medical Transparent IV Cannula Film Fixation Wound Dressing Sterile Waterproof PU Self Adhesive Dressings
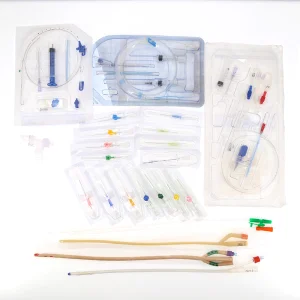 Disposable Medical PVC Latex Silicone Foley Closed Suction Central Venouse Hemodialysis Dialysis IV Cannula Urethral Catheter
Disposable Medical PVC Latex Silicone Foley Closed Suction Central Venouse Hemodialysis Dialysis IV Cannula Urethral Catheter
 Customizable 16-28fr Femoral Dialysis Catheter for Crrt Solutions
Customizable 16-28fr Femoral Dialysis Catheter for Crrt Solutions
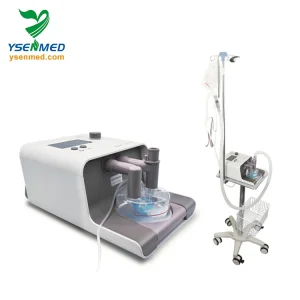 Non-Invasive High Flow Oxygen Therapy System for Hospital Use
Non-Invasive High Flow Oxygen Therapy System for Hospital Use
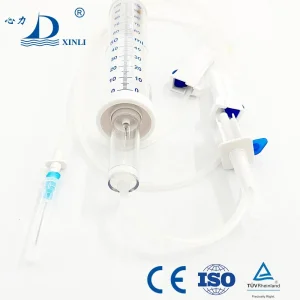
 Disposable Infusion Pump
Disposable Infusion Pump
 Disposable Sterile Medical Infusion Flask for Children
Disposable Sterile Medical Infusion Flask for Children
The global IV Cannula market is currently experiencing an unprecedented era of expansion. As a vital component in modern medical procedures, the IV cannula (intravenous catheter) is indispensable for fluid replacement, blood transfusions, and medication administration. The commercial significance of this sector is underscored by the rising incidence of chronic diseases and an aging global population requiring long-term care. In 2024, the demand for safety-engineered IV cannulas has surged as international health organizations prioritize the reduction of needle-stick injuries among healthcare workers.
From an industrial perspective, manufacturing high-quality IV cannulas requires a sophisticated blend of precision engineering and biocompatible material science. Leading China IV Cannula manufacturers like GreenPlus Medical are at the forefront of this industrial evolution, utilizing medical-grade PTFE (Teflon) or FEP materials to ensure smooth insertion and minimize the risk of phlebitis. The global supply chain now demands products that are not only functional but also compliant with stringent international regulatory frameworks such as the MDR in Europe and FDA guidelines in the United States.
The demand spans across North America, Europe, and emerging markets in Asia-Pacific, with China serving as the primary manufacturing hub for the world.
Passive and active safety mechanisms are now the industry standard to prevent cross-contamination and needle-stick accidents.
Advanced polyurethane (PUR) and FEP materials ensure high flow rates and increased patient comfort during prolonged hospitalization.
The IV cannula industry is no longer just about mass production; it is about intelligent healthcare solutions. Current trends point toward the integration of AI in manufacturing quality control. At GreenPlus Medical, we utilize automated optical inspection (AOI) systems to ensure every needle point is perfectly ground and every catheter tip is tapered with sub-millimeter precision. This technological integration ensures a 99.9% success rate in vein penetration, significantly reducing patient trauma.
Furthermore, the shift toward sustainability is reshaping the industry. Manufacturers are exploring eco-friendly packaging and PVC-free materials to reduce the carbon footprint of disposable medical devices. As a top-tier Chinese supplier, we are leading this transition by optimizing our production lines for energy efficiency and minimizing material waste without compromising sterile integrity.
Years Excellence
Countries Exported
Sterile Guarantee
Global Support
Procurement officers in modern hospitals and clinics look for three critical factors: Reliability, Compliance, and Cost-Effectiveness. The procurement of IV cannulas involves selecting the right gauge (from 14G to 26G) to suit various clinical needs. For instance, in pediatric care, a 24-26G cannula is essential for fragile veins, whereas emergency trauma units require larger 14-16G cannulas for rapid fluid resuscitation.
Localized application scenarios vary greatly. In the European market, there is a heavy emphasis on "Safety Wings" and "Injection Ports" for multi-medication therapy. In contrast, emerging markets may prioritize high-volume procurement of basic models for community health centers. As a versatile China IV Cannula factory, we offer customized OEM/ODM services to meet these localized requirements, providing everything from specialized winged catheters to port-free designs for veterinary or specific clinical uses.
Optimized for ICU, Emergency Rooms, and General Wards with a focus on ease of securement using our proprietary PU dressings.
Strategic proximity to Shenzhen and Hong Kong ports ensures streamlined shipping and reduced lead times for international wholesalers.
Full documentation support including ISO 13485, CE certification, and Free Sale Certificates for smooth customs clearance.
Guangdong GreenPlus Medical Co., Ltd. is a leading professional supplier of high-quality medical instruments and healthcare products based in China. With over 10 years of experience in the medical field, our dedicated team is committed to providing top-notch products and services that enhance the healthcare experience globally.

Located in Guangdong Province, we are strategically positioned near major transportation hubs, including the vibrant city of Shenzhen, allowing us to efficiently serve international markets. Our factory produces a wide range of essential medical products, including syringes, infusion sets, gloves, and face masks. In addition to our in-house manufacturing capabilities, we collaborate with over 5 trusted strategic partners who specialize in producing urine bags, surgical gowns, medical tubes, wound dressings, diagnostic products, and other medical accessories.
At GreenPlus Medical, we place the highest importance on the quality of our products. Our comprehensive Quality Control (QC) team ensures that each production line is carefully monitored, and every product undergoes rigorous testing to meet both international and customer-specific standards. We are proud to hold CE and ISO certifications for many of our products, confirming our dedication to safety and excellence.
We proudly export our products to customers across Europe, Africa, Asia, and America, serving healthcare institutions, distributors, and wholesalers globally. Our robust supply chain ensures timely deliveries, and we continually seek to build lasting relationships with our international partners.
As a China-based IV Cannula manufacturer, we offer more than just competitive pricing. Our advantage lies in the integration of a massive industrial ecosystem found only in the Pearl River Delta. This allows us to source the highest grade of raw materials—from medical-grade stainless steel needles to high-performance polymers—at a scale that ensures consistent quality and cost-efficiency.
Our commitment to healthcare is rooted in the belief that quality care is a fundamental right. We integrate core values of love, respect, and compassion into every product. By choosing Guangdong GreenPlus Medical, you are partnering with a factory that views every IV cannula not just as a commodity, but as a critical tool for saving lives. Our forward-looking approach ensures that we are constantly innovating, developing high-efficiency and professional solutions for the ever-evolving global medical sector.

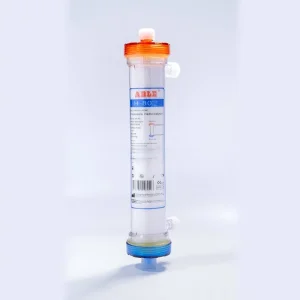 CE ISO Certified Disposable Hemodialyzer Cartridge for Safe Dialysis
CE ISO Certified Disposable Hemodialyzer Cartridge for Safe Dialysis
 Medical Device Needle Free Connector Valve Infusion Injection Adapter Needle Free Injection Connector Infusion Set Needle Free Access
Medical Device Needle Free Connector Valve Infusion Injection Adapter Needle Free Injection Connector Infusion Set Needle Free Access
 Medical Supply Factory Price Eo Sterile Disposable Plastic Positive Pressure Needle Free Connector Valve CE&ISO13485 OEM/ODM, Infusion Use
Medical Supply Factory Price Eo Sterile Disposable Plastic Positive Pressure Needle Free Connector Valve CE&ISO13485 OEM/ODM, Infusion Use
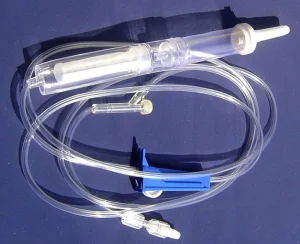 ISO13485 Medical Supplier IV Transfusion Infusion with Blood Filter
ISO13485 Medical Supplier IV Transfusion Infusion with Blood Filter
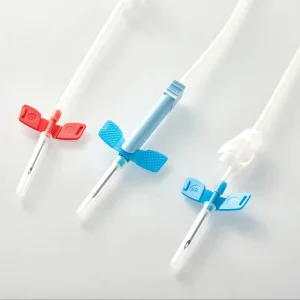 AV. Fistula Needle for Dialysis 15g 16g 17g
AV. Fistula Needle for Dialysis 15g 16g 17g
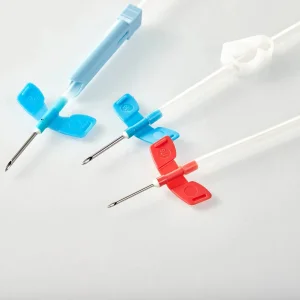 Needle Set IV Fistula for Dialysis Fixed Wing 15g 16g 17g
Needle Set IV Fistula for Dialysis Fixed Wing 15g 16g 17g
 Irrigation Pump Accessories Medical Surgical Accessories
Irrigation Pump Accessories Medical Surgical Accessories
 Disposable Infusion Pump (Advanced Care Model)
Disposable Infusion Pump (Advanced Care Model)