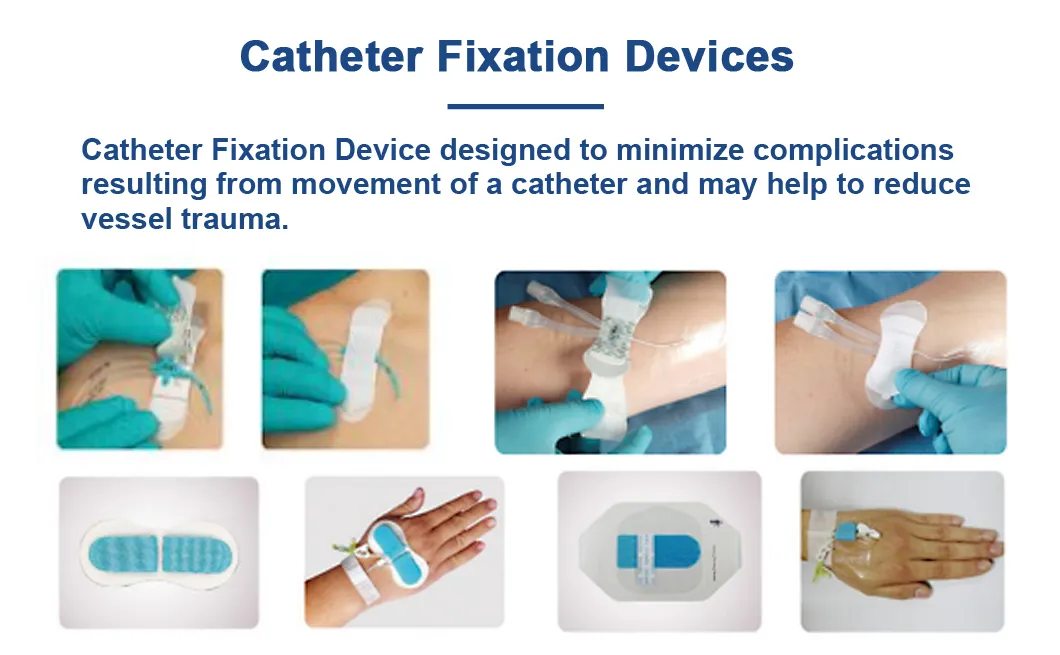
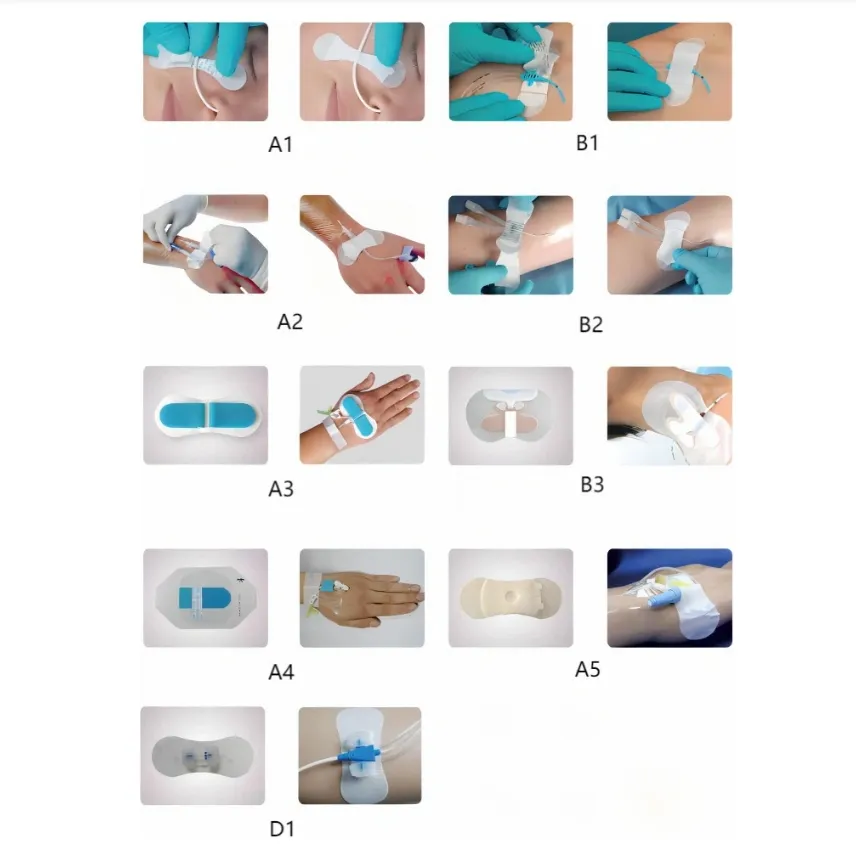
Instructions for Use
1
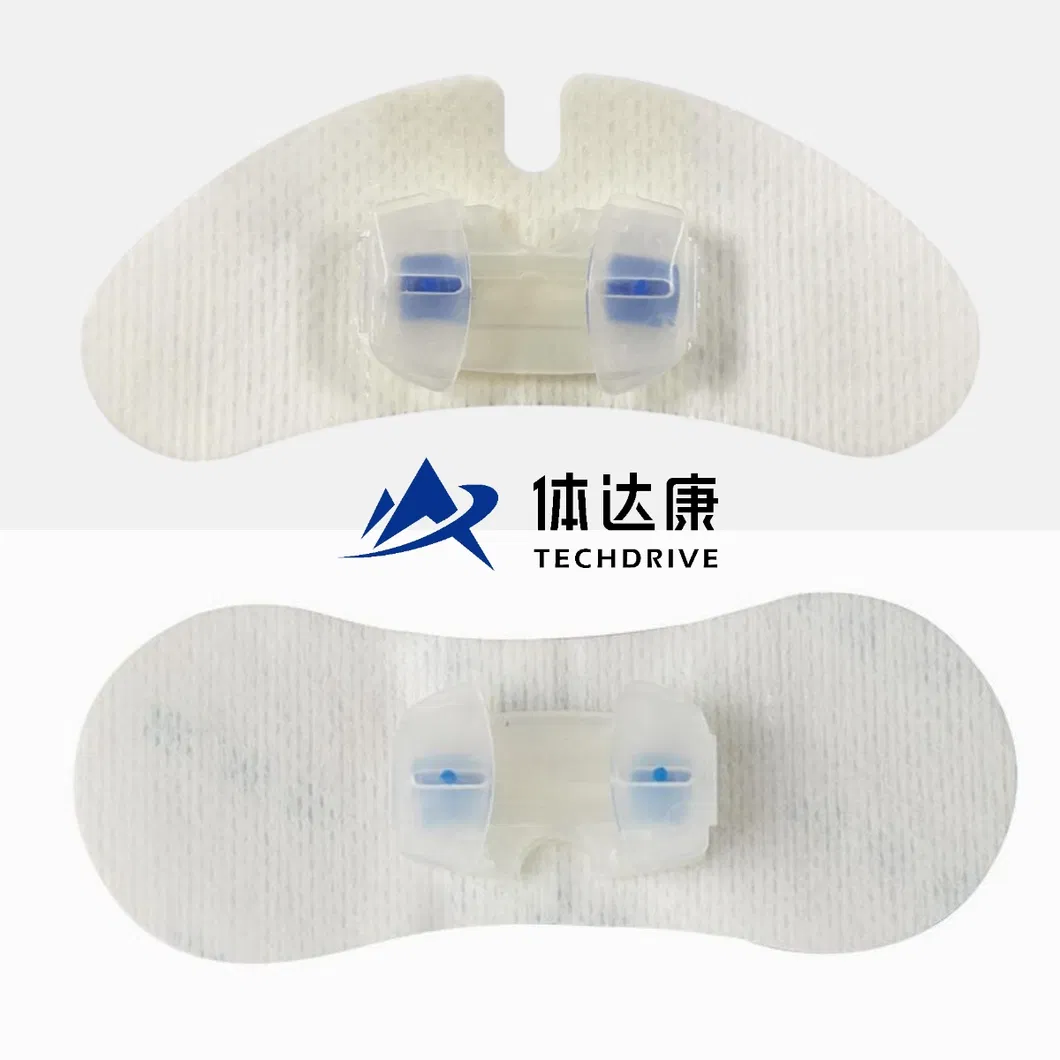
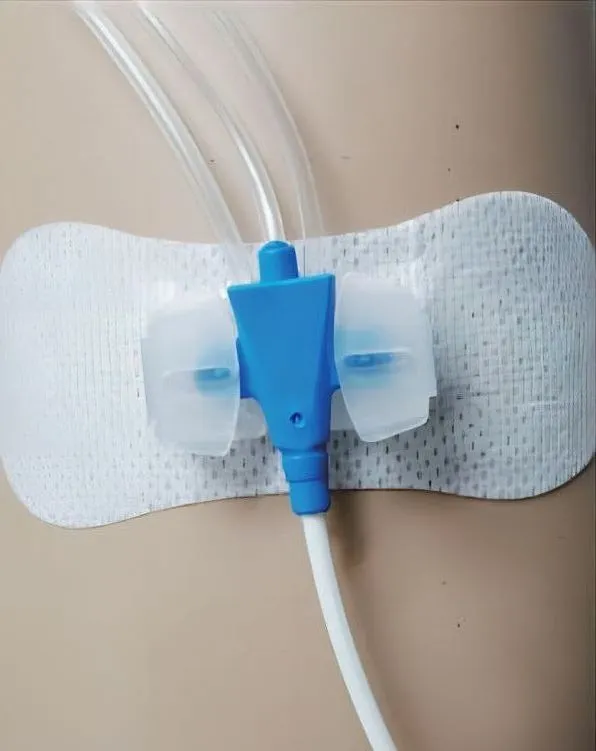
Check the catheter's placement first. Proper placement ensures the rest of the process goes smoothly.
2
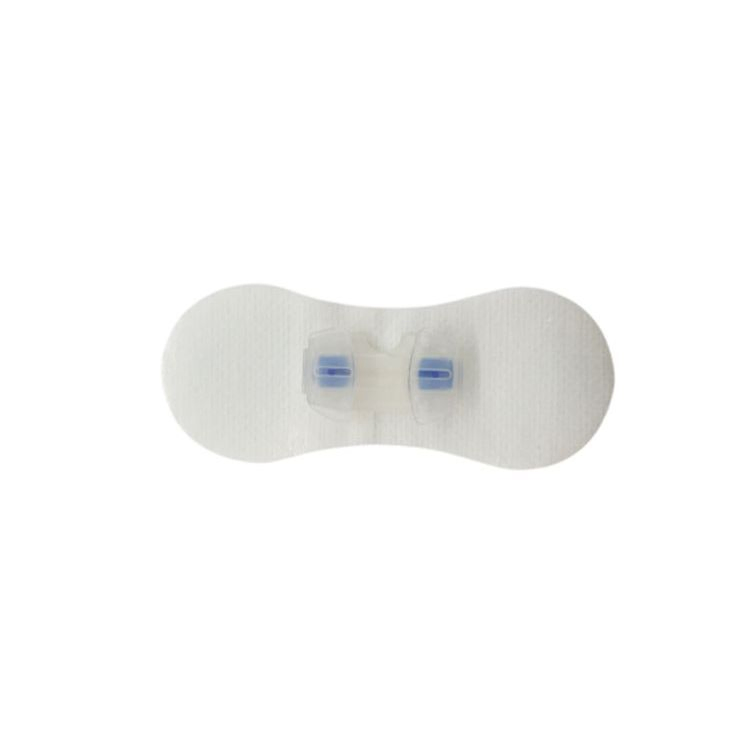
Attach the fixation device to the skin by removing its backing and pressing it firmly around the catheter's entry point. Precision is key for maximum adhesion.
3
Open the plastic fastener upwards, secure the catheter in place, and then close the plastic component to ensure stability.
FAQ
Can you accept OEM or ODM orders?
Yes, we can. Customized orders are also welcomed according to your specific requirements.
How do you control product quality?
We utilize advanced technical equipment and professional talent, adhering to ISO9000 and ISO13485 quality standards to meet and exceed customer demands.
What are your main products?
Our core focus is advanced wound care, specifically catheter fixation devices, silicone foam dressings, alginate dressings, and silicone scar dressings.
What certifications have you obtained?
Our products are certified with FDA, CE, and ISO 13485 to ensure international quality compliance.
What is the nature of the enterprise?
We are a high-tech enterprise integrating research, development, production, and sales of professional medical care products.
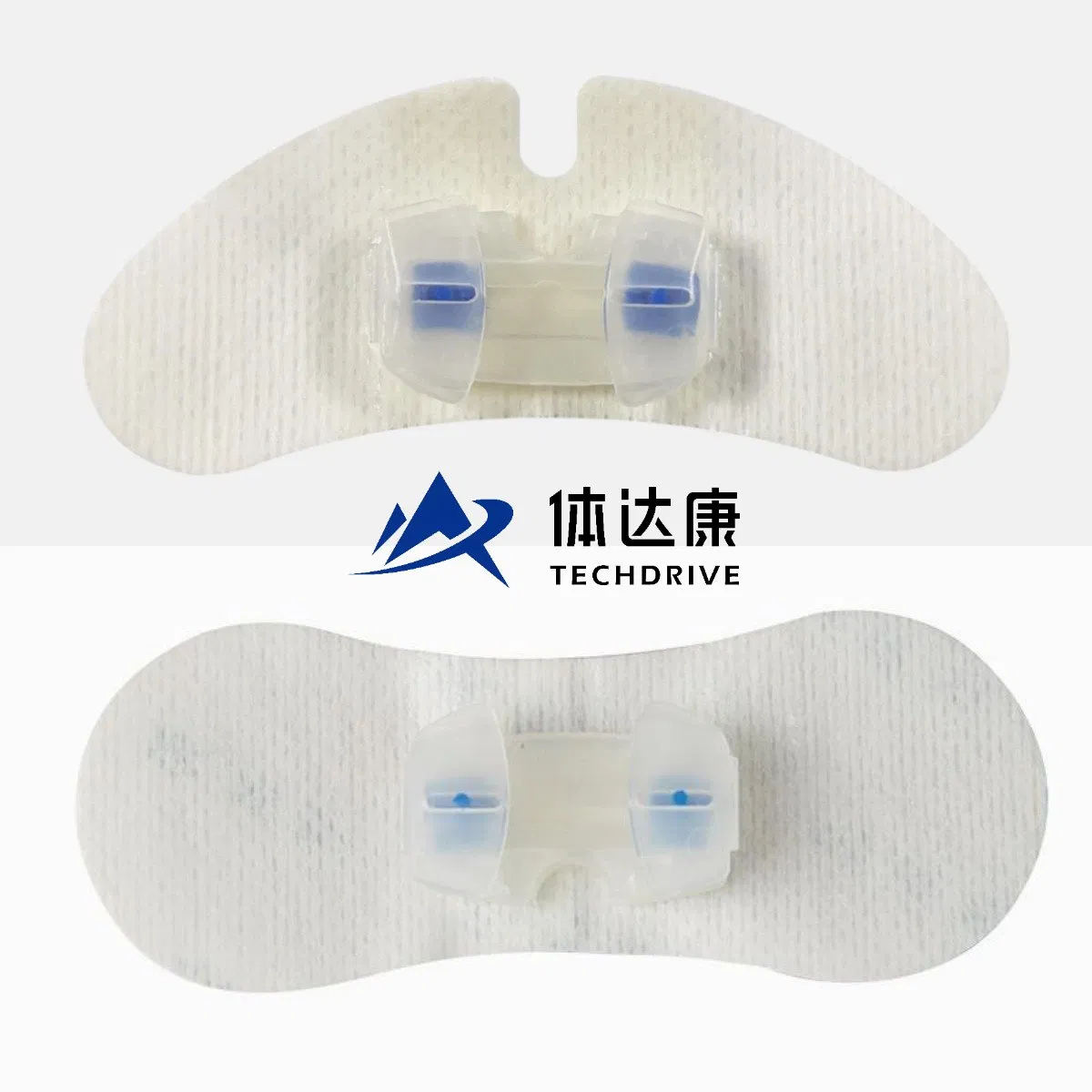
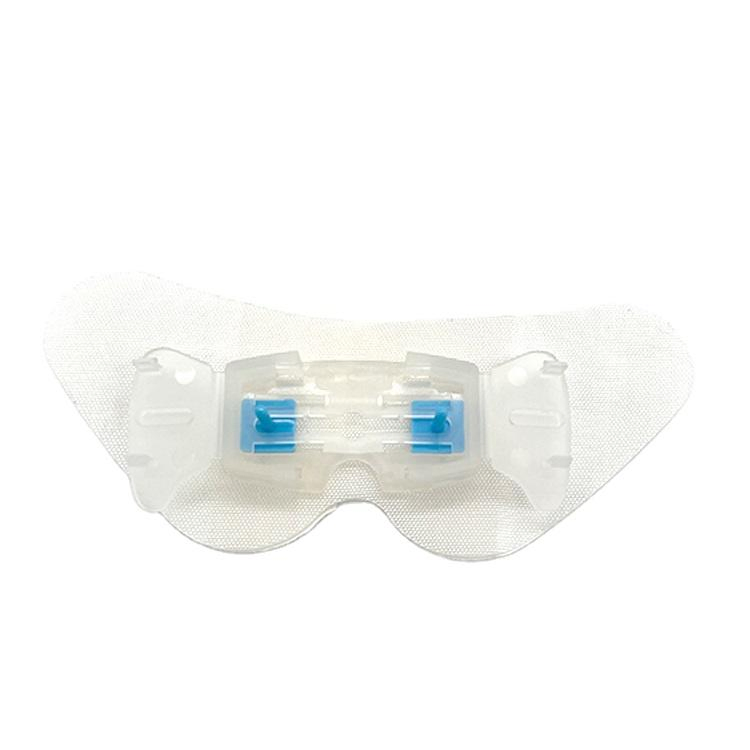


 GreenPlus Medical
GreenPlus Medical