1 / 5
 GreenPlus Medical
GreenPlus Medical










| Still deciding? Get samples of $ ! US$ 0.5/Piece Request Sample |
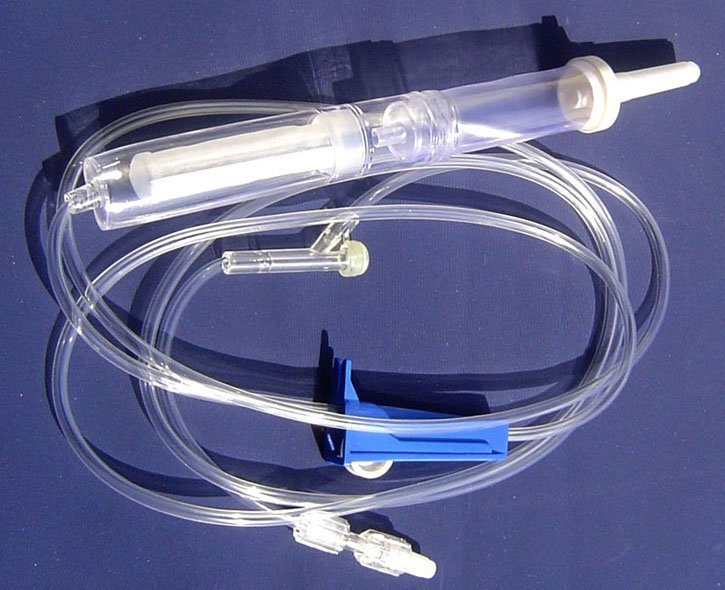
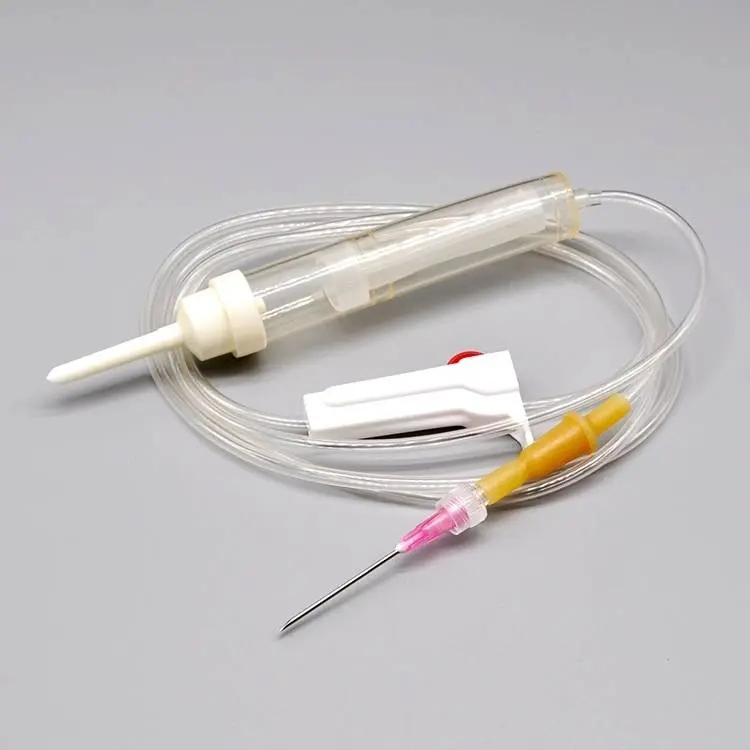
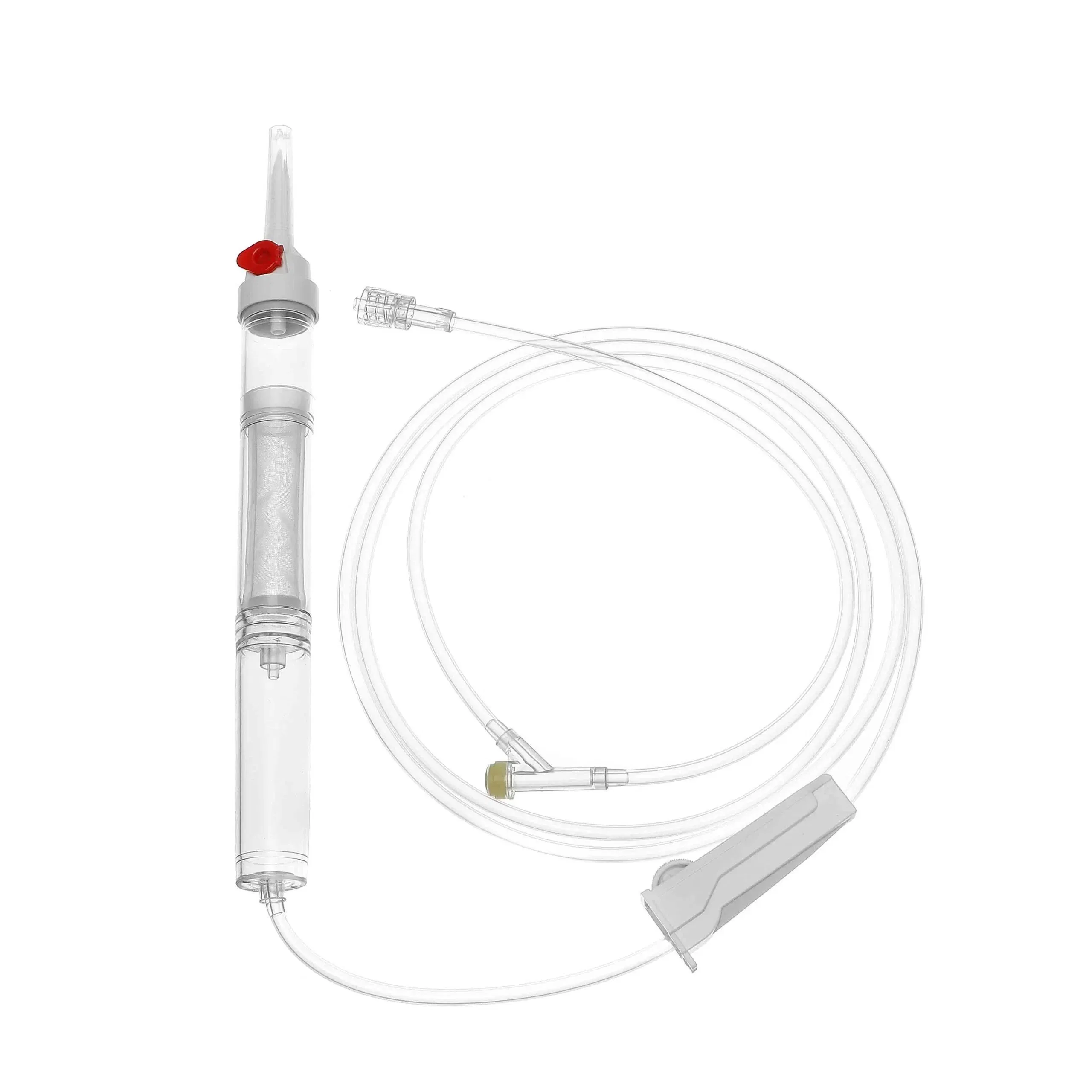
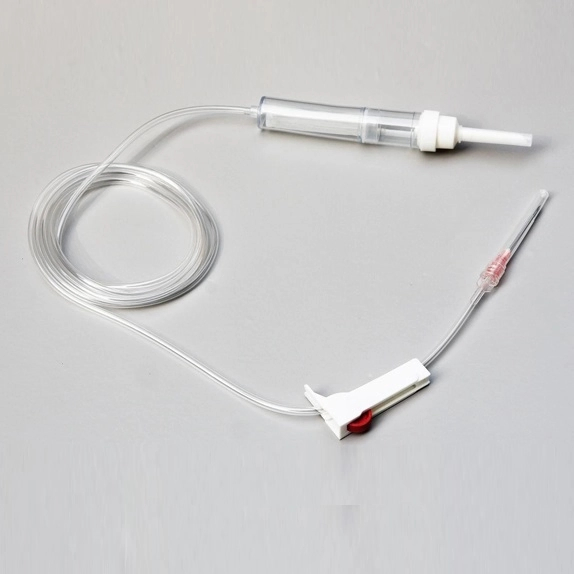
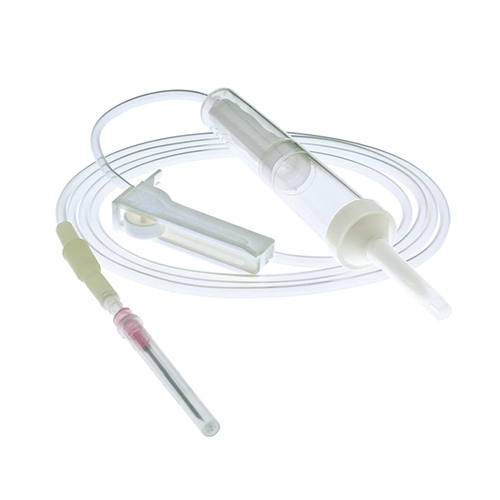
| MATERIAL | ABS, PVC, PP, RUBBER, STAINLESS STEEL |
| PACKING | INNER PE BAG, OUTER CARTON |
| COMPONENTS | Detailed medical components as listed below |
| DIMENSION | 150CM - 200CM |
| LOADING PORT | SHANGHAI PORT, NINGBO PORT, QINGDAO PORT |

